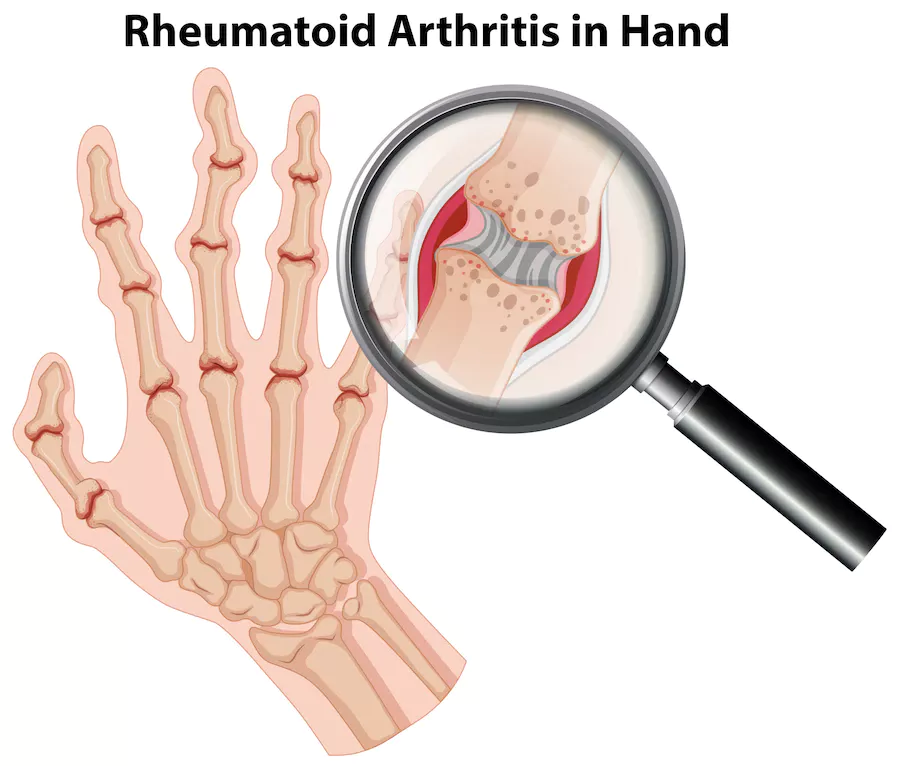
Xeljanz is the brand name for the drug called Tofacitinib. It is used as a medication for the treatment of rheumatoid arthritis, psoriatic arthritis, and ulcerative colitis in adult patients who have been treated with methotrexate without success. It is the first approved medication from JAK (Janus Kinase) inhibitors to treat rheumatoid arthritis. There are no generic alternatives to xeljanz.
Janus kinase (JAK) inhibitors– They are the latest drug class that modulates disease medication to be able to treat rheumatoid arthritis (RA).
Rheumatoid Arthritis– In this disease, the body’s immune system attacks its own tissues causing a chronic inflammatory disorder that affects many joints including the hands and feet.
SIMILARITIES BETWEEN XELJANZ AND XELJANZ XR
- Both are oral tablets i.e. can be taken by mouth. Xeljanz contains tofacitinib while xeljanz xr is an extended-release (long-acting) form of the drug.
- Xeljanz xr also treats rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis like xeljanz.
DOSAGE FOR XELJANZ
This medication can be taken orally once or twice daily with or without food. The initial recommended dose is 5 mg while the recommended dose for xeljanz is 11 mg once daily. The increased doses for xeljanz and xeljanz xr are 10 mg twice a day and 22 mg once a day daily.
EFFECTS AND BENEFITS OF XELJANZ ON THE BODY
- Help to decrease pain, tenderness, and swelling in joints
- Treats some kinds of bowel disease (ulcerative colitis)
- Also helps in reducing the symptoms that occur for ulcerative colitis
- Xeljanz starts working within 3 days
- It is also a non-approved drug by FDA for hair loss after alopecia areata
- It improves healthy hair follicles and encourages hair growth
- Decreases inflammation in the body
Specialty Care Clinics has provided its patient with every required treatment and also it includes the latest approved medication in treatment.
WHO IS RESTRICTED FROM TAKING XELJANZ MEDICATION?
Taking this medication while having the following condition can cause another threat to the body and health.
- Active or inactive tuberculosis
- Opportunistic fungal infection
- Low level of neutrophils (a type of white blood cell)
- A blood clot
- Anemia
- Cancer
- Low level of lymphocytes
- Liver problems and abnormal functioning of the liver
- Bad infection
- A breastfeeding patient
- Chronic kidney disease stage 3A and 3B at a moderate level
- The stomach wall or intestine ruptured
- Malignant lymphoma
- Chronic kidney disease stage 4 (severe) and stage 5 (failure)
- Opportunistic infection
WHAT TO AVOID DURING XELJANZ
There are certain things you should avoid while on medication.
- Grapefruit juice (strictly prohibited)– It is not at all recommended to take grapefruit juice with tofacitinib. Because it increases both tofacitinib levels as well as the risk of side effects. Follow the advice from your healthcare professional.
- Drinking excessive alcohol– Although there is no warning regarding the consumption of alcohol it is advised not to consume too much alcohol as it may raise obstacles in treatment.
FOOD AND DRUG APPROVAL FOR XELJANZ (TOFACITINIB)
Its approval history started in 2012 and the last approval was given in 2021 by FDA
- In 2012, the xeljanz tablet was approved to be consumed orally.
- In 2016, the extended-release was xeljanz xr, for oral consumption
- In 2020, the xeljanz oral solution
- In 2021, the tofacitinib citrate tablet was approved
- The same year in 2021 (August), the tofacitinib tablet extended release
FEW QUESTIONS PEOPLE ASK RELATED TO XELJANZ:
Most common side effects:
- High blood pressure
- Fever
- Headache
- Nausea, vomiting, or diarrhea
- Runny nose, sneezing, and sore throat
- Skin rashes or shingles
How long does it take for xeljanz to work?
Usually, xeljanz starts its work within 3 days of starting and provides significant results in ulcerative colitis symptoms in the form of improvement. The induction 1 and 2 of phase III Octave trials also showed significant improvement in ulcerative colitis symptoms within two weeks.
Xeljanz medication’s generic availability
Xeljanz is available under brand name only. Its generic form is still not available. It contains an active drug called Tofacitinib.
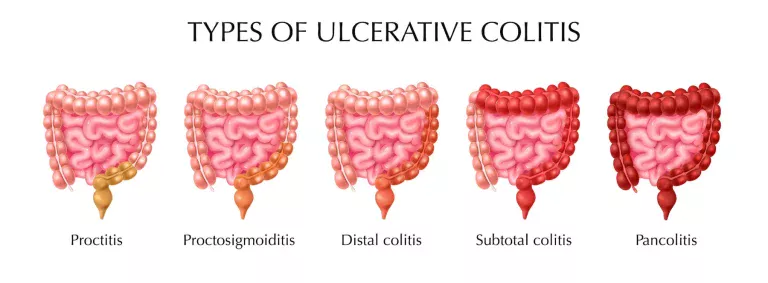
Xeljanz’s effectiveness on ulcerative colitis
As we read above that xeljanz and xeljanz xr both (as FDA approved) treat ulcerative colitis (from moderate to severe) in adults. The American College of Gastroenterology has specified some guidelines for treatment.
Xeljanz (Tofacitinib) as an active drug treats patients who didn’t receive enough benefits of TNF inhibitors or got any side effects. At Specialty Care Clinic, we treat as well as provide initial to last dose recommendations.
To book an appointment call us at (469)545-9983.